POLARIXPARTNER Management Consultancy
Are you looking for a consultancy that recognizes the potential of your company and realizes it together with you? Do you want to know which adjustments you need to make in order to remain competitive?
POLARIXPARTNER is your implementation-oriented management consultancy which supports you in perfectly exploiting the performance potential of your company. We guide you with customized solutions and support you position your company for a brighter future.
Together, we find ways and solutions that are tailored to you and your needs. Through the comprehensive know-how and the many years of experience of our consulting team, we seize opportunities to put you at the top of your industry. With our extensive expertise in the core industries of automotive, aerospace, medical technology, the pharmaceutical industry, as well as in the plant construction and energy sectors, we are here to assist you with thorough guidance.
Let us find solutions to overcome the current and upcoming challenges you are facing!
With comprehensive expertise, we accompany you as you embrace major future trends: e-mobility, autonomous driving and digitalization. We look forward to talking to you.
Topic of the month
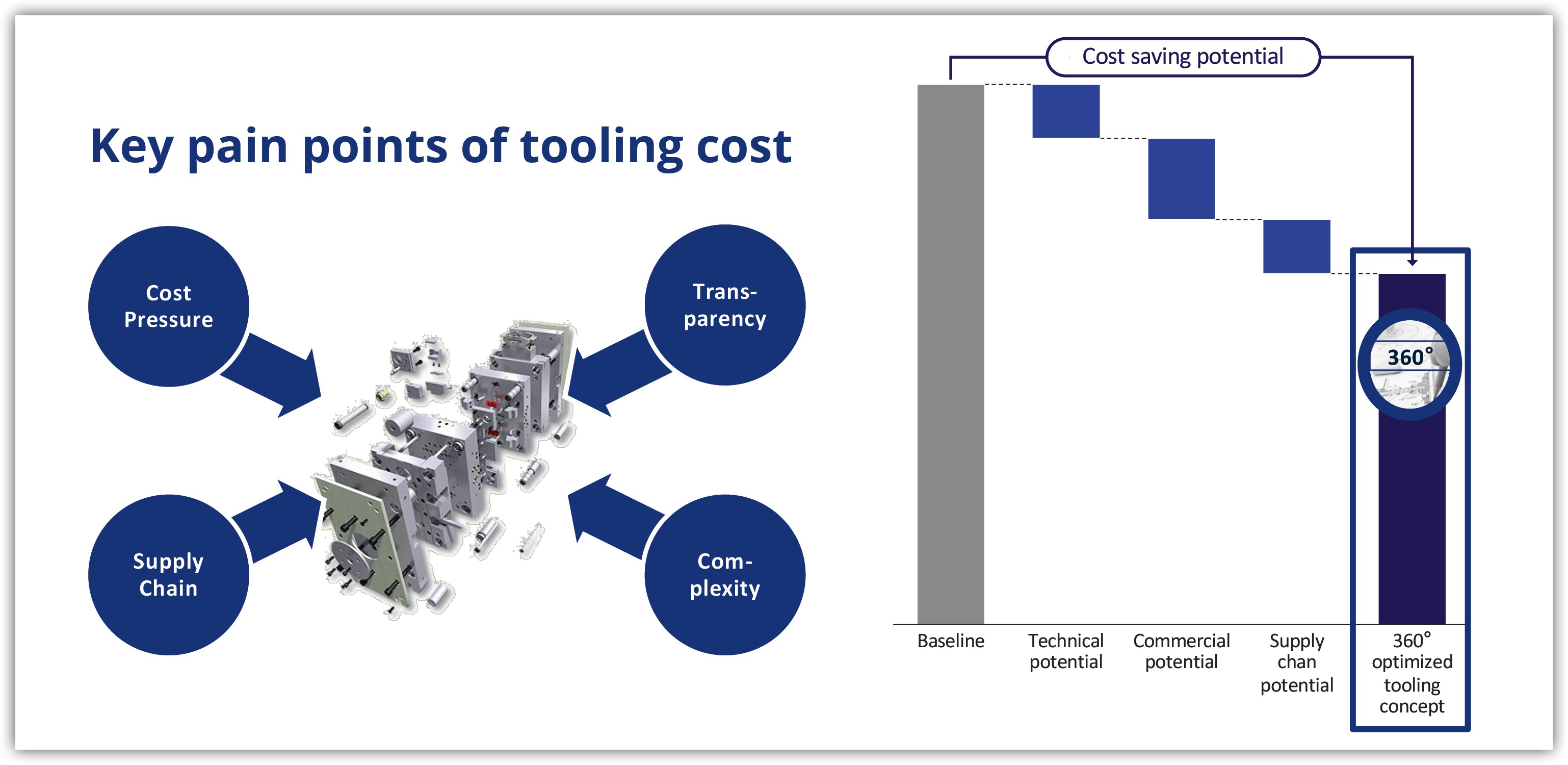
🔧 Key Pain Points in Tooling Costs:
➡️ Raw material, energy and labor costs are highly volatile
➡️ Inflation-driven cost escalation creates uncertainty in long-term tooling projects
➡️ Tooling prices often negotiated as package without detailed breakdowns
➡️ Limited internal reference data for complex tool designs
➡️ Continuous shift of tool manufacturing to Eastern Europe and Asia (China, Turkey, India)
➡️ Reduced planning reliability resulting from increased coordination complexity
➡️ Growing part complexity and hybrid processes for lightweight or composite parts
➡️ Toolmaker capacity and market pressure as critical influencing factor
📊 Our Solution:
✔ Definition of the optimal technical tooling concept by validating design feasibility, liminating over-engineering, and optimizing tool layout, process flow, and material selection
✔ Generation of full cost transparency through detailed should cost analysis, benchmark-based evaluations, and negotiation support to achieve the best price-performance ratio.
✔ Identification of the most competitive sourcing regions and suppliers by balancing cost, quality, and lead time - ensuring reliable global tool supply and reduced risk.
👉 How is your organization dealing with cost volatility? Let’s exchange experiences and best practices!
Your team at POLARIXPARTNER
COST ENGINEERING OF THE FUTURE – AI-SCAN OF PCBAs
Together with our partner K|Lens, we will present our unique AI-scanner of electronic platines (PCBAs), a cutting-edge technology for the next generation of cost engineering. Kaleidoscopic camera snaps high resoluted 3 dimensional pictures from 9 perspectives within one shot. Witness a fully automated 3-D scan system that can detect all semiconductors and microcontrollers, transferring them directly into a costed BOM and a CO2-Footprint!
The appetizer is here. Watch out our video.
Automotive Insiders: Supplier RFQ Timing and Cost Calculations
The automotive industry is in a dramatic change. The main topics are e-mobility, autonomous driving & new mobility concepts. Innovation cycles will shorten significantly, and as a consequence, the product life cycles will also shrink. The share of Software in a car will increase while the percentage of mechanics goes down. A car might be perceived as a software device.
Many automotive suppliers have expressed concerns that their OEM customers have expectations for quote response times that don’t allow for adequate profitability analysis. In the Podcast, Markus Wiederstein (POLARIXPARTNER) and John Monica (Siemens) discuss RFQ process improvement supported by a new cost calculation tool.
Our competencies for your success!
We use our consulting expertise and technical know-how to find the solutions to meet your individual challenges. POLARIXPARTNER offers you real added value and helps you change the future in the following areas:
PRODUCT DEVELOPMENT & PRODUCT OPTIMIZATION
Design-to-cost
Design-to-value
Requirements management (risk management, change management, implementation management)
Lean Development
Cost-efficient development, SCRUM
SUPPLY CHAIN MANAGEMENT
Supply Chain Planning
Global network planning (best-cost country)
Global Manufacturing Footprint
Optimization of the Value Chain
Benefit from our industry know-how
POLARIXPARNTER is more than just a management or strategy consultancy: we don't just advise you, we actively solve problems! Our consultants combine the key competences of a management consultancy with many years of operational industry experience.
In more than 700 successful projects, we have proven that we are at your side as a partner for the operational implementation of the identified optimization. We act as drivers of change and provide the necessary impulse for your continuous change process. Our consultants support you with strategic vision and comprehensive expertise from an average of 16 years of experience in the industry as well as in the consultancy.
Our values in consulting
Our company name POLARIXPARTNER always corresponds to our guiding principle in every step we take with you: We are your reliable guiding star, accompanying you in your search for individual business solutions. You set the direction and we lead you to the goal!
Our consulting services consist of a balanced consulting concept and a holistic approach to your company. In doing so, we dive deep into your core processes analytically and strategically and also become active directly on the shop floor to ensure optimal improvements along your entire value chain.
We offer industry expertise and problem-solving concepts as an added value for you. We support you with industry-specific know-how, sound methodological knowledge and a high degree of social competence. Hand in hand, we develop efficient strategies and forward-looking concepts for your company - fast, results-oriented, in line with targets and realization-oriented.
POLARIXPARTNER Insights
With POLARIXPARTNER Insights, we offer you independent evaluations of trends and developments. In our Insights, we cover the latest topics and trends and share our point of view. For example, would you like to know where the future of your industry is going, and how to make the most out of upcoming market opportunities? We provide you with forward-looking white papers, publications and studies! Whether it's the automotive, aerospace, medical technology or pharmaceutical industry, mechanical engineering or plant construction – we have the inside knowledge to help you with your strategic development.





